Demystifying Quantum Numbers: Understanding Their Role in Quantum Mechanics
What is Quantum Number? Types, Examples & Their Significance
Quantum numbers are numerical values that describe the unique quantum state of an electron in an atom. These numbers help us understand the arrangement of electrons and the structure of atoms, which is critical for chemistry, physics, and modern technologies like quantum computing.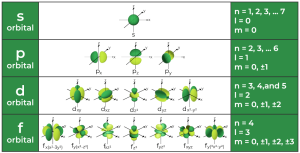
Types of Quantum Numbers
There are four main quantum numbers, each representing different properties of electrons:
1. Principal Quantum Number (n)
- Denotes the main energy level (shell) of an electron.
- Values: n = 1, 2, 3, …
- Larger
nmeans the electron is farther from the nucleus.
2. Angular Momentum Quantum Number (l)
- Describes the shape of the orbital.
- Values: l = 0 to (n – 1)
- For example, if n = 3, then l = 0, 1, 2 (representing s, p, d orbitals)
3. Magnetic Quantum Number (ml)
- Specifies the orientation of the orbital in space.
- Values: ml = –l to +l
- If l = 1 (p orbital), ml can be –1, 0, or +1.
4. Spin Quantum Number (ms)
- Indicates the direction of the electron’s spin.
- Values: +½ or –½
- Important in explaining electron pairing and magnetic properties.
🔄 Quantum Number Example
For an electron in the 3p orbital:
- n = 3 (third energy level)
- l = 1 (p orbital)
- ml = –1, 0, or +1 (any one value, depending on electron)
- ms = +½ or –½ (depends on spin)
📌 Why Quantum Numbers Matter
- Electron configuration: Determines how electrons fill atomic orbitals.
- Chemical behavior: Influences reactivity, bonding, and periodic trends.
- Quantum computing: Forms the theoretical basis for quantum bits (qubits) and algorithms.
💡 What is the MS Quantum Number (Spin)?
The MS quantum number, or spin quantum number, explains the intrinsic angular momentum of an electron — basically, which direction it spins.
- Possible values: +½ or –½
- Electrons in the same orbital must have opposite spins (Pauli Exclusion Principle).
- Spin quantum numbers explain magnetism and electron pairing.
🧠 Bonus Section: Quantum Numbers in Quantum Computing
Quantum numbers don’t just live in textbooks — they power the next-gen tech revolution.
In quantum computing, the spin of particles like electrons and atomic nuclei can serve as qubits (quantum bits), the fundamental units of quantum computers. Understanding these quantum properties allows engineers and physicists to build machines that simulate atoms, break cryptography, and solve complex problems faster than classical computers.
Thanks for reading! Let us know in the comments if you’d like a full beginner’s guide to quantum computing next!
